 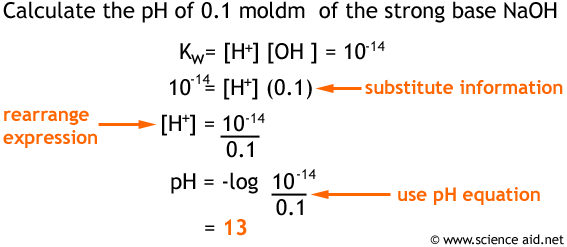
The “server” ( server.R) that responds to user interface events in the web browser.The user interface ( ui.R) that controls the sliders and passes inputs to the model.A script ( carbonate.R) that performs the simple carbonate equilibrium calculations In the analysis of acid-base chemistry, the Henderson-Hasselbalch equation and the Stewart strong ion formulation can only describe (rather than predict) the.This website is controlled using the R package How this website works (including all the code!) Dutkiewicz (2006), On the solution of the carbonate chemistry system in ocean biogeochemistry models, Ocean Modelling, 12(3-4), 290–301, doi:10.1016/j.ocemod.2005.05.004. 
Princeton University Press, 2006.įollows, M. Self-ionization of water occurs only to a small amount in aqueous solutions of strong acids and strong bases. Follows, Ocean Dynamics and the Carbon Cycle. Given below is the pH calculation formula: k w ( H 3 O ) ( O H ) 1.0 × 10 14 p K w p H p O H 14 Strong Acids and Strong Bases Strong acids and strong bases in dilute aqueous solutions basically complete their ionization. (ed), “ Environmental Chemistry,” Oxford University Press, 1998. Tans, “Why Carbon Dioxide from Fossil Fuel Burning Won't Go Away” In: MacAladay, J. You can also download, share as well as print the list of Chemistry calculators with all the formulas. These calculators will be useful for everyone and save time with the complex procedure involved to obtain the calculation results. To read the code, click the “ Website Code” tab to the right. A tool perform calculations on the concepts and applications for Chemistry calculations. The iterative solution and then backsubstitution is a lot simpler than it sounds. The pH and pOH Calculator is useful when working with chemicals. This means that doubling atmospheric pCO\(_2\) (a 100% increase) would lead to only a 10% increase in marine DIC. The Revelle factor indicates the fractional change in pCO\(_2\) per fractional change in DIC, and is about 10 for current global mean conditions. Determining pH and Kw We can determine the hydrogen ion concentration in water from measuring its pH. Water Chemistry Calculator Size Of Pool 20,000 Gallons Salt pH Total Alkalinity Cyanuric Acid Hardness Phosphates Before adding salt, test your pool water for the current level of. It is also useful to calculate the “Revelle buffer factor,” which is approximately Perfect pool chemistry should keep pH between 7.4 and 7.6, meaning the pool should be slightly more basic than acidic. The calculation is based on simplified carbonate equilibria including dissolved CO \(_2\), bicarbonate ion, ,, and pCO\(_2\). This web app calculates the partial pressure of CO \(_2\) (pCO \(_2\)) and pH of seawater in equilbrium with the atmosphere given sea-surface temperature, titration alkalinity, and dissolved inorganic carbon. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
